In order to evaluate its application as a dental prosthesis material, a CoCrW alloy was subjected to in vitro cytotoxicity test, surface characterization and electrochemical studies performed in artificial saliva and 0.15 mol.L-1 NaCl medium. The used techniques were: anodic polarization curves, chronoamperometric measurements, electrochemical impedance spectroscopy (EIS), scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy (EDS) analysis and X-ray photoelectron spectroscopy (XPS). Cytotoxicity test was also performed. The electrochemical behavior of CoCrW alloy was compared in both studied media, from corrosion potential (Ecorr) to a 600 mV anodic overvoltage. From the electrochemical measurements it was observed that the CoCrW alloy in both media presents only generalized corrosion. SEM and EDS analysis showed that the alloy presents carbide niobium and silicon and manganese oxides as nonmetallic inclusions. XPS results indicated that cobalt does not significantly contribute to the passivating film formation. Cytotoxicity test showed no cytotoxic character of CoCrW alloy. These results suggest that the CoCrW alloy can be used as biomaterial to be applied as prosthesis in dental implants.
Introduction
Metal alloys have been used since the beginning of the 20th century as material in dentistry due to their mechanical properties (1,2). Nowadays, due to economic issues, non-precious alloys are replacing noble alloys in dentistry. Dental non-precious metal alloys present a thin passive film of oxide on the surface. This passive film must have high adhesion, be compact, present high electrical resistance and absence of defects such as cracks (1,2).
The oral environment is perfect to promote the oxidation of metallic materials. Factors like quantity and quality of saliva, salivary pH, dental plaque, amount of protein, chemical and physical properties of food and fluid intake, and general oral health conditions may influence the metal corrosion in the oral cavity (3). There are two concerns about dental materials in oral environment: the localized effects or systemic damage caused by the release of corrosion products to the body and the effects on the physical properties and clinical performance of the alloy (4-7).
CoCr-based alloys have been utilized in dental prosthesis, since they offer good resistance to corrosion (8,9). Recently, alloys with high content of tungsten instead molybdenum have been developed aiming a better ceramic- metal adhesion (10). On the other hand, the CoCrMo alloys have been more studied (4-12) than CoCrW alloys (13,14).
In this work, in vitro cytotoxicity test, surface characterization and electrochemical behavior of a CoCrW alloy have been performed in artificial saliva and 0.15 mol.L-1 NaCl medium in order to evaluate its application as dental prosthesis material. For characterizing the alloy surface, polarization curves, chronoamperometry, electrochemical impedance spectroscopy (EIS), scanning electron microscopy (SEM) , energy dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectroscopy were used. In vitro cytotoxicity test was also performed to study the biocompatibility of the proposed alloy as implant material.
Material and Methods
Sample and Solutions
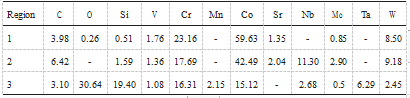
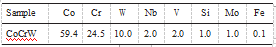
The chemical composition of the CoCrW alloy is in Table 1.
The compositions of the artificial saliva were prepared from three different solutions. The following reagents/concentrations (mol.L-1) were used: solution A:NaH2PO4/0.233 + KCl/1.164 + NaCl/0.123 + NH4Cl/0.205+ sodium citrate/3.74x10-3 + lactic acid/0.039; solution B- urea/0.167 + uric acid/4.46x10-3 + NaOH/5x10-3 and solution C: KSCN/0.123. The saliva solution was prepared daily by mixing A, B and C solutions (1:1:1) and then diluted 50 times in high-purity de-ionized water (15).
Table 1. Chemical composition of CoCrW alloy (wt )
All the experiments were performed in triplicate with naturally aerated solution at (37.0±0.5) °C and a 6.6 pH (oral conditions).
Electrodes
The CoCrW alloy working electrodes were discs fabricated from the central part of the bars and had an area of 0.90 cm2. A cylindrical epoxy-base was fitted with the steel disk. A concentric brass rod was coupled to the steel+epoxy-base. The electrodes were prepared by polishing with successively finer grades of emery papers of 120, 400, 600 and 2000 mesh and then thoroughly rinsed with distilled water and ethanol and air dried prior to the experiments.
The auxiliary electrode consisted of a platinum foil and it was cleaned with acid and flamed just before each experiment. Saturated calomel electrode (SCE) was used as reference electrode.
Electrochemical Experiments
The corrosion resistance was evaluated by potentiodynamic polarization anodic curves and chronoamperometric curves. The studied CoCrW alloy was in artificial saliva and 0.15 mol.L-1 NaCl solutions. These evaluations were performed after the attainment of a constant current at each potential application, starting from the open circuit potential in the anodic direction from Ecorr till 0.9 V vs. SCE using 1 mV.s-1 scan rate. As working criterion, it was assumed that the transpassivation potential is reached when the current density is 10 mA/ cm2. The electrochemical impedance spectroscopy (EIS) measurements were performed over eight frequency decades (from 100 KHz to 10 mHz). The potential was ±8 mV, p.p. The experiments were conducted at 37 °C. A
μAutolab type III/FRA2 potentiostat (Metrohm Autolab BV, the Netherlands) was used coupled to a frequency response detector and to a microcomputer.
Table 2. Different surface regions analyzed using energy dispersive spectroscopy (EDS) for CoCrW alloy after polishing